 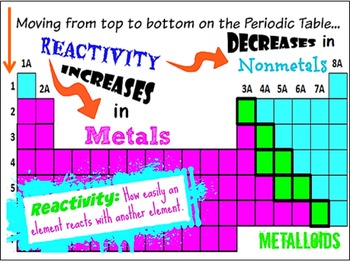
It increases on moving from left to right as more energy is released. The affinity of electrons – refers to the energy that is released when an electron is gained by an atom. On moving in a diagonal direction, atomic radius tends to increase. On moving from left to right, it increases, and on moving from top to bottom, it increases. Ionisation energy increases on moving horizontally across a period, whereas on moving in the downward direction, the ionisation energy decreases.Ītomic radii – the distance between the outermost electron and the nucleus is known as the atomic radius. Ionisation energy – this energy helps the atoms to remove the electrons. An exception in group 3 elements is present where electronegativity increases towards thallium. On moving in the downward direction, the electronegativity decreases. On moving from left to right horizontally in a periodic table, there is an increase in electronegativity. The Pauling scale is used to measure electronegativity. Some periodic trends and chemical reactivity in the periodic table are as follows –Įlectronegativity – measures the ability of a molecule to gain electrons within a chemical bond. The trends of the elements in a periodic table depend on various factors like electronegativity, ionisation energy, electron affinity, atomic radii, metallic character, and chemical reactivity. Periodic trends refer to the patterns of the properties of the chemical elements. The periodic trends and chemical reactivities of elements are shared below. The gas elements do not gain or lose their electrons because the electronic configuration is filled. Hence the elements on the left side in the downwards direction are the highly reactive elements. In a period, the chemical reactivity decreases on moving towards the right, whereas in a group, chemical reactivity increases on moving downwards. Hence the electronegativity and the ionisation energy determine a chemical reaction. The higher the reactivity of the element, the easier it is to combine. The electrons between the elements are shared, or the elements lose or gain the electrons. Therefore, as you go down the group the outermost electron becomes easier to lose and the reactivity increases as you go down the group.The rate at which a chemical substance or an element undergoes a chemical reaction is known as reactivity. Therefore, the attraction of the outer electron to the nucleus decreases as you go down group 1. This increases the shielding effect the positively charged nucleus has on the outer electron. The number of electron shells increases.The electrostatic attraction of the outer electron to the positive nucleus decreases as you go down group 1. Therefore, the outer electron is further away from the positive nucleus, as you go down the group. This is due to the number of shells increasing. Explaining the trend in reactivityĪs you go down the group the reactivity of the Group 1 increases because: 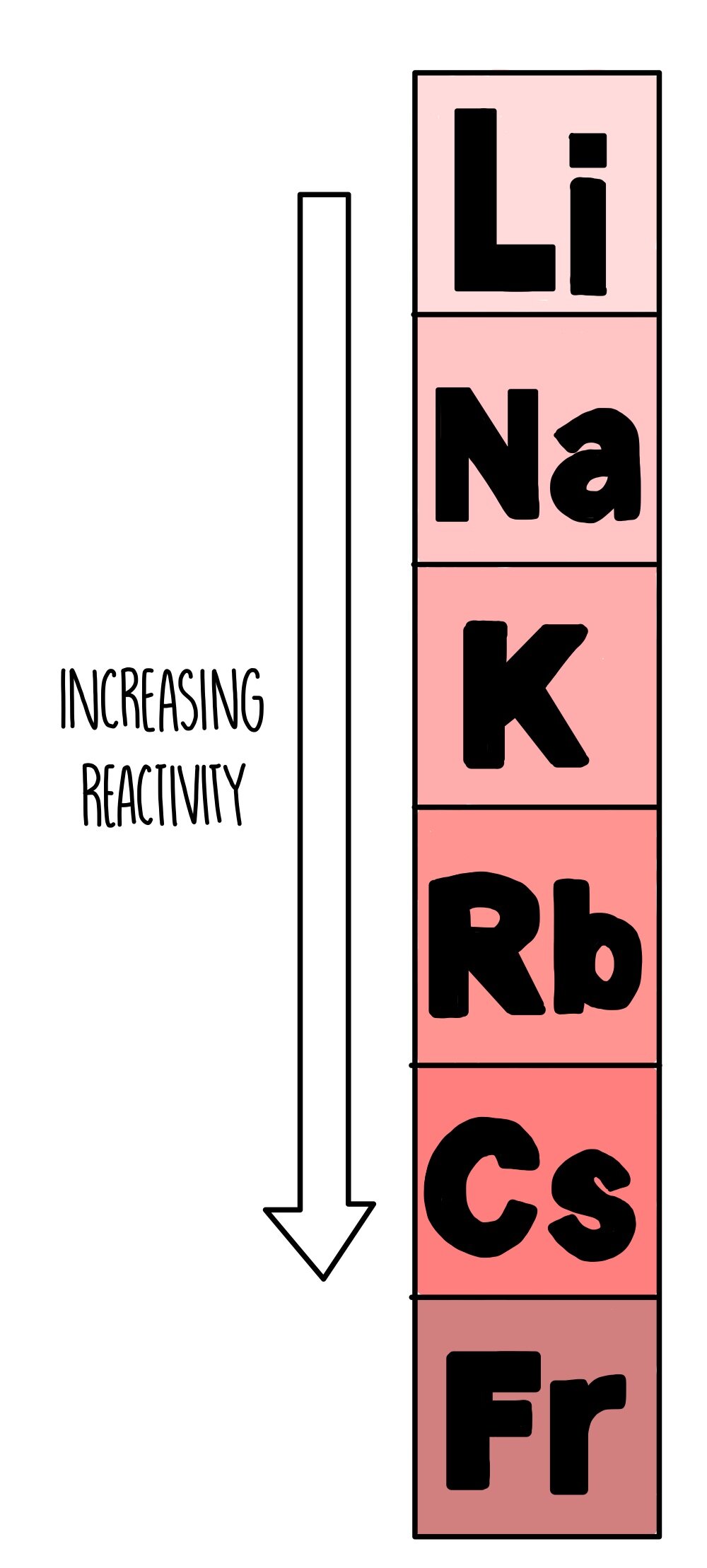
So lithium would be the least reactive and francium would be the most reactive out of all the elements in group 1. This suggests that as you go down group 1, the reactivity of the elements increases. Looking at the reactions discussed beforehand, when going down group 1 the reactions get more and more vigorous. Trend in reactivity in a group can be explained using the electronic structure of atoms. Group 1: Reactivity (GCSE Chemistry) Group 1: Reactivity Group 1 – trend in reactivity
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
